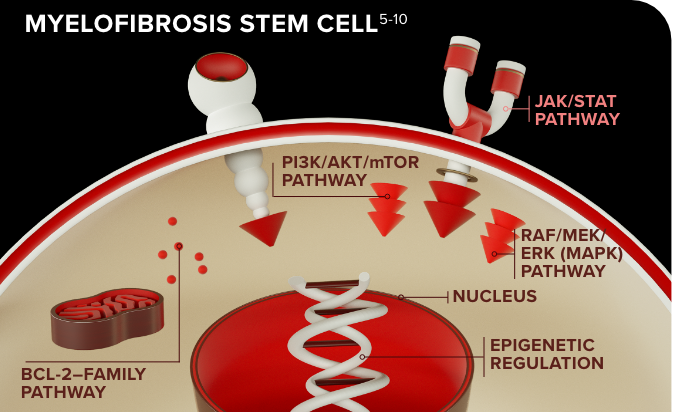
Multiple pathways drive myelofibrosis progression1
Pathways include JAK/STAT, BCL-2 family, epigenetic regulators, PI3K, and RAF/MEK/ERK (MAPK)1,2
- Targeting the downstream signaling may still permit malignant stem cells to evade apoptosis and the disease to progress2,3
- Additional pathways allow the continued survival of malignant clones that drive disease progression3,4
AKT=protein kinase B; BCL-2=B-cell lymphoma 2; BCL-XL=B-cell lymphoma-extra large; ERK=extracellular-signal–regulated kinase; JAK/STAT=Janus kinase signal transducer and activator of transcription; MAPK=mitogen-activated protein kinase; MCL-1=myeloid cell leukemia-1; MEK=MAPK/ERK kinase; MF=myelofibrosis; mTOR=mammalian target of rapamycin; PI3K=phosphoinositide 3-kinase; RAF=rapidly accelerated fibrosarcoma.
References:
- Pettit K, Odenike O. Novel therapies for myelofibrosis. Curr Hematol Malig Rep. 2017;12(6):611-624. doi:10.1007/s11899-017-0403-0
- Schieber M, Crispino JD, Stein B. Myelofibrosis in 2019: moving beyond JAK2 inhibition. Blood Cancer J. 2019;9(9):74. doi:10.1038/s41408-019-0236-2
- Santos FPS, Verstovsek S. Therapy with JAK2 inhibitors for myeloproliferative neoplasms. Hematol Oncol Clin North Am. 2012;26(5):1083-1099. doi:10.1016/j.hoc.2012.07.008
- Gleitz HFE, Pritchard JE, Kramann R, Schneider RK. Fibrosis driving myofibroblast precursors in MPN and new therapeutic pathways. HemaSphere. 2019;3(S2):142-145. doi:10.1097/HS9.0000000000000216
- Ishida S, Akiyama H, Umezawa Y, et al. Mechanisms for mTORC1 activation and synergistic induction of apoptosis by ruxolitinib and BH3 mimetics or autophagy inhibitors in JAK2-V617F-expressing leukemic cells including newly established PVTL-2. Oncotarget. 2018;9(42):26834-26851. doi:10.18632/oncotarget.25515
- Gozgit JM, Bebernitz G, Patil P, et al. Effects of the JAK2 inhibitor, AZ960, on Pim/BAD/BCL-xL survival signaling in the human JAK2 V617F cell line SET-2. J Biol Chem. 2008;283(47):32334-32343. doi:10.1074/jbc.M803813200
- Tremblay D, Mascarenhas J. Next generation therapeutics for the treatment of myelofibrosis. Cells. 2021;10(5):1034. doi:10.3390/cells10051034
- Feng Y, Chen X, Cassady K, et al. The role of mTOR inhibitors in hematologic disease: from bench to bedside. Front Oncol. 2021;10:611690. doi:10.3389/fonc.2020.611690
- Meyer SC, Levine RL. Molecular pathways: molecular basis for sensitivity and resistance to JAK kinase inhibitors. Clin Cancer Res. 2014;20(8):2051-2059. doi:10.1158/1078-0432.CCR-13-0279
- Moens U, Kostenko S, Sveinbjørnsson B. The role of mitigen-activated protein kinase-activated protein kinases (MAPKAPKs) in inflammation. Genes. 2013;4(2):101-133. doi:10.3390/genes4020101
- Yang Q, Crispino JD, Wen QJ. Kinase signaling and targeted therapy for primary myelofibrosis. Exp Hematol. 2017;48:32-38. doi:10.1016/j.exphem.2016.12.007
- Agarwal A, Morrone K, Bartenstein M, Zhao ZJ, Verma A, Goel S. Bone marrow fibrosis in primary myelofibrosis: pathogenic mechanisms and the role of TGF-β. Stem Cell Investig. 2016;3(5):1-10. doi:10.3978/j.issn.2306-9759.2016.02.03
- Petiti J, Lo Iacono M, Rosso V, et al. Bcl-xL represents a therapeutic target in Philadelphia negative myeloproliferative neoplasms. J Cell Mol Med. 2020;24(18):10978-10986. doi:10.1111/jcmm.15730
- Tognon R, Gasparotto EPL, Neves RP, et al. Deregulation of apoptosis-related genes is associated with PRV1 overexpression and JAK2 V617F allele burden in essential thrombocythemia and myelofibrosis. J Hematol Onc. 2012;5(2):1-11. doi:10.1186/1756-8722-5-2
- Rubert J, Qian Z, Andraos R, Guthy DA, Radimerski T. Bim and Mcl-1 exert key roles in regulating JAK2V617F cell survival. BMC Cancer. 2011;11:24. doi:10.1186/1471-2407-11-24
- Mughal TI, Vaddi K, Sarlis NJ, Verstovsek S. Myelofibrosis-associated complications: pathogenesis, clinical manifestations, and effects on outcomes. Int J Gen Med. 2014;7:89-101. doi:10.2147/IJGM.S51800
- Harrison CN, McLornan DP. Current treatment algorithm for the management of patients with myelofibrosis, JAK inhibitors, and beyond. Hematology Am Soc Hematol Educ Program. 2017;(1):489-497. doi:10.1182/asheducation-2017.1.489
- Kramann R, Schneider RK. The identification of fibrosis-driving myofibroblast precursors reveals new therapeutic avenues in myelofibrosis. Blood. 2018;131(19):2111-2119. doi:10.1182/blood-2018-02-834820